Properties of water
Water plays a critical role in botany due to its unique properties that support plant life and physiological processes. Below are the key properties of water relevant to botany:
### 1. **Polarity and Solvent Properties**
- Water is a polar molecule, meaning it has a partial positive charge on the hydrogen atoms and a partial negative charge on the oxygen atom. This polarity allows water to dissolve many substances, especially ionic and polar compounds.
- As a result, water acts as a universal solvent in plants, facilitating the transport of nutrients, minerals, and other essential substances through the plant's vascular system.
### 2. **High Heat Capacity**
- Water has a high specific heat capacity, meaning it can absorb or release a large amount of heat without undergoing significant temperature changes.
- This property helps regulate temperature within plant cells and tissues, providing a stable internal environment even when external temperatures fluctuate.
### 3. **High Heat of Vaporization**
- Water requires a significant amount of energy to transition from liquid to gas (vaporization). This property is crucial for transpiration, the process by which water evaporates from the leaves of plants.
- Transpiration helps cool the plant and creates a "pull" that draws water up from the roots to the leaves.
### 4. **Cohesion and Adhesion**
- Cohesion refers to the attraction between water molecules, which contributes to surface tension and the formation of water droplets. In plants, cohesion helps maintain the continuity of the water column in the xylem.
- Adhesion is the attraction between water molecules and other surfaces, such as the walls of xylem vessels. This property, combined with cohesion, enables capillary action, allowing water to move upward against gravity in plants.
### 5. **States of Water**
- Water exists in three states: solid (ice), liquid (water), and gas (water vapor). In plants, water is primarily in the liquid state, but it can also be found as vapor in the process of transpiration.
- The unique property of water being less dense as a solid allows ice to float, which is important in natural water bodies, but less directly relevant to plant physiology.
### 6. **pH and Buffering Capacity**
- Water can dissociate into hydrogen ions (H⁺) and hydroxide ions (OH⁻), making it a key component in pH regulation. The pH of water affects the availability of nutrients in the soil and the health of plant cells.
- Buffers in plant systems help maintain a stable pH, which is essential for enzyme activity and other biochemical processes.
### 7. **Transport and Capillary Action**
- Water’s cohesive and adhesive properties facilitate capillary action, which is vital for the transport of water and dissolved minerals from the roots to the leaves.
- This process is essential for photosynthesis and the overall growth and development of plants.
### 8. **Surface Tension**
- Surface tension, a result of cohesive forces, allows water to form droplets and resist external forces. This property is important for the movement of water across plant surfaces and in small vessels.
### 9. **Hydration and Cellular Functions**
- Water is essential for maintaining the turgor pressure in plant cells, which is necessary for structural support and growth.
- It also participates in various biochemical reactions, including photosynthesis, respiration, and nutrient transport.
### 10. **Role in Photosynthesis**
- Water is a critical reactant in the light-dependent reactions of photosynthesis, where it is split into oxygen, protons, and electrons. This process provides the electrons needed to produce ATP and NADPH, which are used in the Calvin cycle to synthesize glucose.
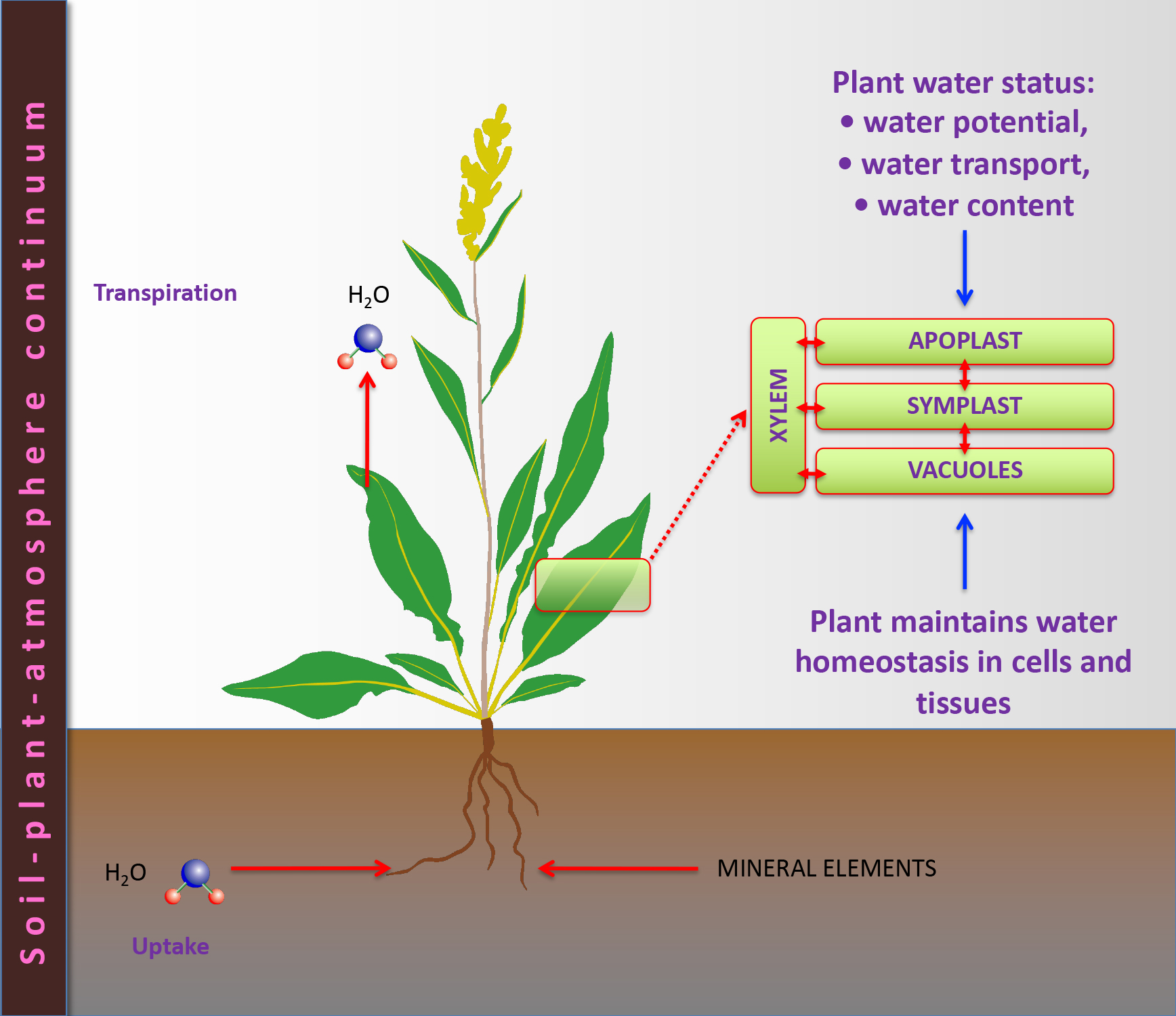
*Water journey in plants: from roots to transpiration.*
These properties collectively ensure that water supports the structural, physiological, and biochemical needs of plants, making it indispensable for plant life and growth.